Genome Architecture
The genome stores, propagates and expresses genetic information. Delineating the genome architecture is the prerequisite to dissect fundamental DNA-templated processes including transcription, replication, repair.
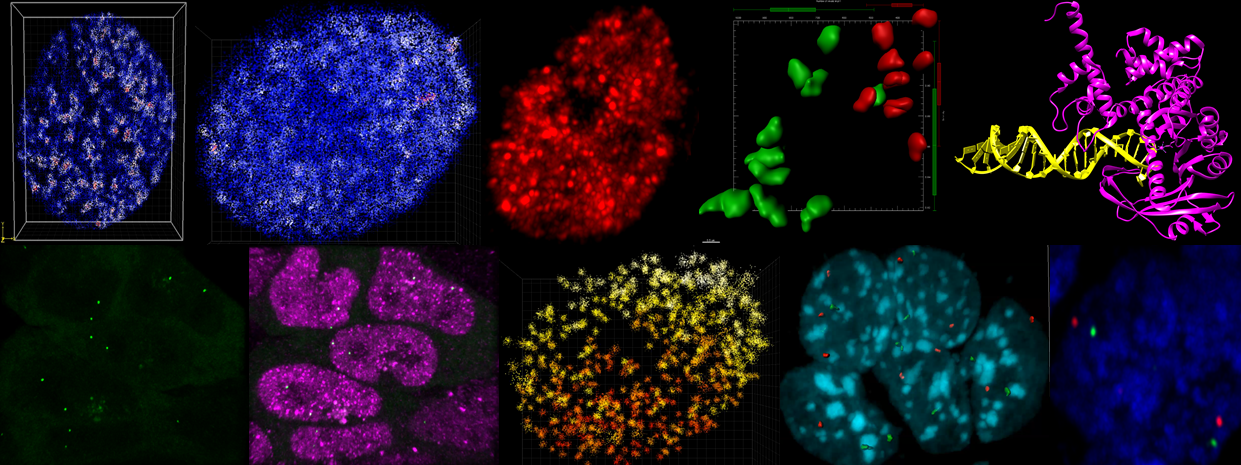
We previously developed a new super-resolution imaging method, 3D ATAC-PALM, to selectively capture the spatial organization of DNA regulatory elements (e.g., enhancers, promoters, insulators) in single cells in situ at nanometer resolution (Xie et al., Nature Methods, 2020). The Xie lab will continue to build new molecular and computational tools to deconvolve the mammalian genome.
Genome Regulation
3D ATAC-PALM discovers that the accessible genome, comprising key DNA regulatory elements, are non-homogeneously organized into spatial clusters we called accessible chromatin domains. The mechanism and function of these spatial clusters are poorly defined.
Combining imaging, genomics and simulation, we identified the bromodomain and extraterminal (BET) family protein BRD2 as a novel regulator of the clustered organization of the accessible genome (Xie et al., Nature Genetics, 2022) . Dysregulation of BRD2 has been found in cancer, developmental disorders, immuno-inflammatory and infectious diseases (e.g., COVID19). The Xie lab will combine cutting-edge imaging, genomics and computation to dissect the mechanisms by which BRD2 regulates the genome structure and function underlying human diseases.
Demystifying Enhancer
Among various types of cis-regulatory DNA elements within the accessible genome, enhancers could work over long-genomic distance (kilo- to mega-base) relative to gene promoters and activate developmental stage- and tissue-specific gene transcription program. Dysregulation of enhancers can cause human developmental defects and diseases. How distal enhancers organize in space and time to communicate with target promoters remains a central mystery of human transcription regulation.
We previously defined a long-range enhancer cluster (~50-70kb) driving the Yamanaka reprogramming factor Klf4 gene expression and interplaying with the highly dynamic yet intrinsically ordered pluripotency transcription factors ensemble by live-cell single-molecule imaging, genome engineering and in vitro biochemistry (Xie et al., Genes & Development, 2017). The Xie lab will build novel molecular and computational tools to dissect enhancer-guided gene regulation in development and disease, with surprising observations and novel mechanistic insights continuing to unfold.
Decoding Cancer Genome
In addition to common structural variations (e.g., insertion, deletion, translocation) of the cancer genome, oncogenes and their regulatory elements could transform into excessively large (~ megabases), highly amplified (~hundreds of copies), circular DNA structures, called extrachromosomal DNA (ecDNA). ecDNAs promote massive oncogene expression, drive tumor heterogeneity and evolution, associate with drug resistance and poor clinical outcome. Therefore, ecDNA has been designated as a Cancer Grand Challenge.
In collaboration with Howard Chang and Paul Mischel at Stanford, we discovered that ecDNAs spatially cluster into hubs that drive high level oncogene expression (King*, Yost*, Xie* et al, Nature, 2021). At Cleveland Clinic, the Xie lab will develop new microscopy and analytical tools to study the dynamics and regulation of ecDNA in cancer.